Since our foundation in 1717(Kyoho 2nd year of the Edo period), we have fully committed to the pharmaceutical business, under the corporate philosophy “Dedicated to the Fight against Disease and Pain”. In FY2021, we have newly established sustainable management policy, to realize a sustainable society.

| Vision over the medium- to long-term | |
|---|---|
| 1 |
|
| 2 |
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
|
| 7 |
|
| 8 |
|
| 9 |
|
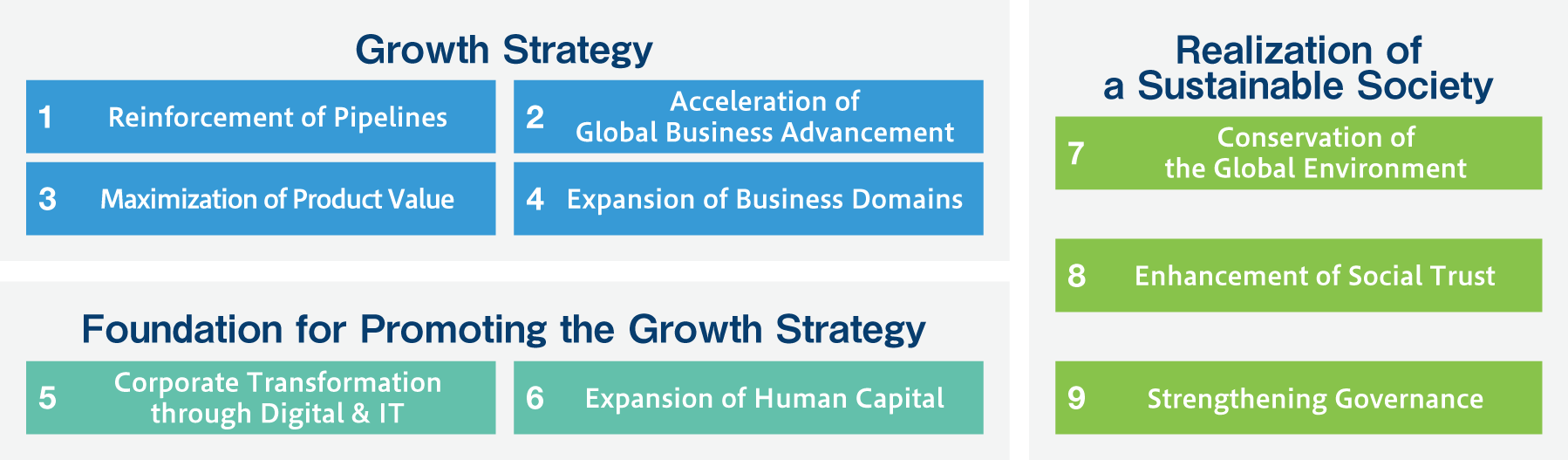
In FY2021, based on the newly established sustainable management policy, we changed the materiality from “important CSR issues” to “important management issues” to analyze and manage financial and non-financial management issues in more integrated way. The materiality thus defined has been clearly linked to the strategy of the mid-term management plan and has been developed into a more dynamic management system.
We believe that the disclosure of integrated financial and non-financial information and dialogues will be possible so that stakeholders outside of ONO can understand our sustainability initiatives.
In June 2024, Deciphera Pharmaceuticals, Inc. (in the United States), joined the Ono Pharmaceutical Group, enabling direct sales in Europe and the United States. This prompted us to review and consider our materiality. This revision does not change the materiality identification process that we conducted in fiscal 2021. In March 2025, we reclassified the 18 materiality into 9 items based also on our continued dialogue with institutional investors.
The materiality analysis implemented in FY2021 was conducted using the following process, which is updated annually as progress is checked.


In the materiality analysis, we conducted a management environmental analysis in conjunction with the formulation of the mid-term management plan to extract potential management issues. This analysis identified important opportunities and risks for creating value and achieving sustainable growth of our company. Our directors, executive officers, and senior management from all divisions participated in the external/internal management environmental analysis, which included analysis of the management environment surrounding the business and analysis of gaps between our long-term vision and current status. In addition, management issues were extracted based on requests and expectations of stakeholders that were confirmed by each division in its daily business activities. As for non-financial issues, we extracted issues related to intangible assets such as human capital and intellectual capital that are needed to realize our growth strategies.
Non-financial issues were updated based on ISO 26000, the GRI Standards, the SASB Standards, the Ten Principles of the United Nations Global Compact, evaluations by ESG-rating agencies, dialogues with investors, etc. Analysis of issues was conducted while the progress of deliberation was reported to and confirmed by the Board of Directors.
In identifying materiality in 2021, we first classified the issues extracted in Step 1 into “value creation,” “foundation for value creation,” or “value preservation (erosion risks).” We recognized that “value creation” and “foundation for value creation” are opportunities and “value preservation” is a risk for our company. Furthermore, at the Management Meeting and other occasions, 18 materiality issues* were defined as the most important issues from the perspective of importance to stakeholders and business. Later in June 2024, Deciphera Pharmaceuticals, Inc. (in the United States), joined the Ono Pharmaceutical Group, enabling direct sales in Europe and the United States. This prompted us to review and consider our materiality. This revision does not change the materiality identification process that we conducted in fiscal 2021. In March 2025, we reclassified the 18 materiality into 9 items based also on our continued dialogue with institutional investors. Materiality issues were deliberated and finalized by the Board of Directors. Based on the content of ongoing dialogue with institutional investors, in March 2025, we consolidated the 18 materiality items into 9 items from the dual perspectives of "importance to the environment and society" and "importance to our company."
Please see the “Actions for Materiality Issue” for reason for being a priority issue, targets and progresses for each materiality.
In regard to the comprehensive materiality analysis in FY2024, we engaged in a dialogue with external experts about the process of our materiality analysis, the themes that are set and future initiatives to verify the validity of each important issue.
With regard to each materiality issue that was redefined in FY2021, we will establish mid-term targets and plans, and confirm the progress. In addition, these are extracted using enterprise risk management (ERM) and are managed in an integrated manner with managed risks (click here for more information on ERM).
We establish our company-wide PDCA management cycle by linking KPIs and progress for each materiality to our medium-term management plan, as well as by linking them to corresponding divisions, organizations, and committees. The KPIs and progress for each materiality are managed by the Board of Directors and the Executive Committee. We also conduct periodical reviews and analyze external/internal environments every year to review progress against materiality issues and medium- and long-term objectives.
In accordance with the annual review conducted in FY2023, the names and contents of some materiality items were changed. In March 2025, we reclassified the 18 materiality into 9 items based also on our continued dialogue with institutional investors.Please see below for reason for being a priority issue, targets and actions for each materiality and the progress by year.
ONO has striven to develop our CSR by defining important areas of focus based on ISO 26000. In FY2018, we redefined our materiality as “important CSR issues” to clarify CSR activity themes that we should emphasize. ONO is actively engaged in CSR in accordance with the materiality that we have established.
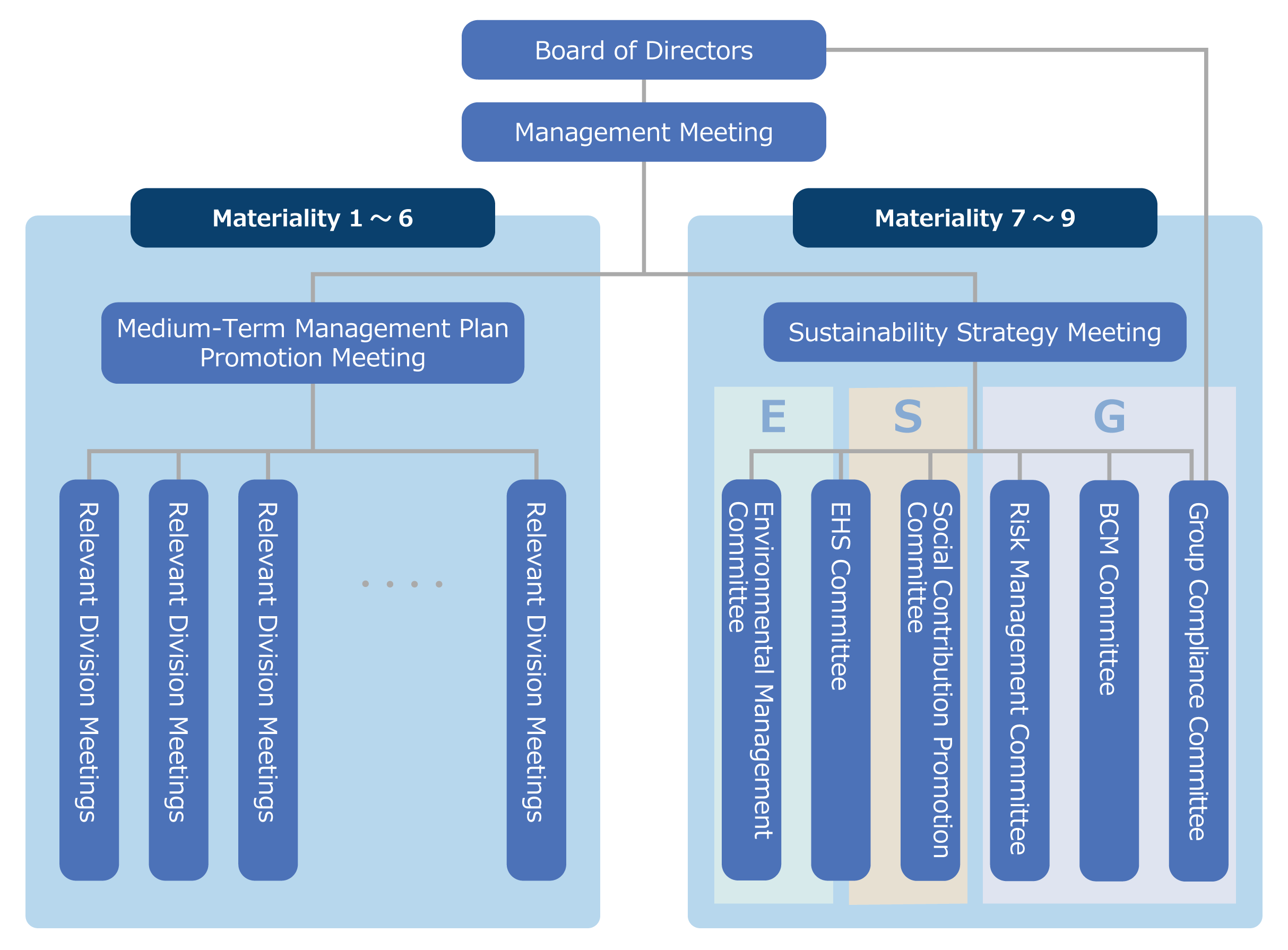
At Ono Pharmaceutical, the Board of Directors oversees important management issues (materiality) in sustainable management, and appoints the Representative Director, President & COO as the chief Sustainability Management Officer, and the Representative Director, Executive Vice President as the director in charge of sustainability.
Under the Representative Director, President & COO, the Sustainability Strategy Meeting (which is chaired by the director in charge of sustainability and consists of the Representative Director, President & COO, the Executive Directors, auditors, and the headquarters office manager determined by the chair) has been established to discuss and deliberate important matters. The Sustainability Strategy Committee, together with the six committees shown below, has established a corporate governance system that closely cooperates with the Board of Directors.
In November 2017, we participated in the United Nations Global Compact (UNGC), which is composed of 10 principles advocated by the UN concerning human rights, labour, environment, and anti-corruption. We comply with relevant laws and disseminate “the Ten Principles of the UNGC” through our daily activities to ensure that all employees follow them.

We submit our Communication on Progress (CoP) every year to the UNGC to report on our initiatives toward “the 10 Principles of the UNGC.”

We contribute to Goal 3, Goal 9 and Goal 17 in the SDGs through the creation of innovative drugs.

We strive to realize Goal 3: Ensure healthy lives and promote well-being at all ages as a research and development company specializing in prescription drugs based on our corporate philosophy to be dedicated to the fight against disease and pain. In response to the mortality rate of non-communicable diseases raised as a goal of the SDGs, we began to concentrate our research area into diseases such as cancers, immunological diseases and central nervous system disorders to contribute to the creation of original and innovative therapeutic medications for diseases for which medical needs have still not yet been satisfied. To improve access to healthcare in low-income and low- and middle-income countries, we will work in partnership with NGOs and other organizations to strengthen healthcare systems over the medium to long term, including the development of medical personnel and the improvement of healthcare environments.

In terms of Goal 9: Build resilient infrastructure, promote inclusive and sustainable industrialization and foster innovation, ONO contributes to encouraging innovation and building research and development infrastructure. To vitalize research and development in order to create new drugs, we of course not only invest in internal research and development but also provide grants, such as those for investigator-initiated clinical trials. Furthermore, the ONO Medical Research Foundation and ONO Pharma Foundation promote research to help build a bedrock for innovation through research grants to researchers overseas.

Moreover, we cannot separate ourselves from the duty to promote innovation or from Goal 17: Strengthen the means of implementation and revitalize the global partnership for sustainable development. We will not only provide innovative drugs independently but also seek out and achieve a wide range of partnerships. Long before “open innovation” became a commonly used phrase, ONO advanced the development of new drugs through the use of state-of-the-art technology and expertise from various fields worldwide. At the same time, we have been actively working to introduce and draw on new candidate compounds for pharmaceuticals. In addition to alliances with venture companies and other pharmaceutical companies, we form partnerships with a wide range of stakeholders from universities and research institutes to government agencies, local communities and NPOs in an effort to resolve problems via open innovation. A list of our main partnerships can be found here.
