In today’s society, in which mass production and mass consumption continue to expand along with global economic growth and increases in population, pollution of the natural environment and damage to ecosystems related to the disposal of waste have become problems, while at the same time it is projected that our limited resources may run dry. In consideration of this situation, we have set Realization of a Resource Recycling Society as one of the major items of our medium-to-long term ECO Vision for implementing our business activities, and have been promoting a variety of initiatives on a company-wide basis.
In order to realize a resource recycling society, we have been working to achieve the following goals since FY2023.
| Final Landfill Disposal Rate of Industrial Waste |
|
|---|---|
| Recycling Rate |
|
| Reduce the Environmental Impact of Product Packaging |
|
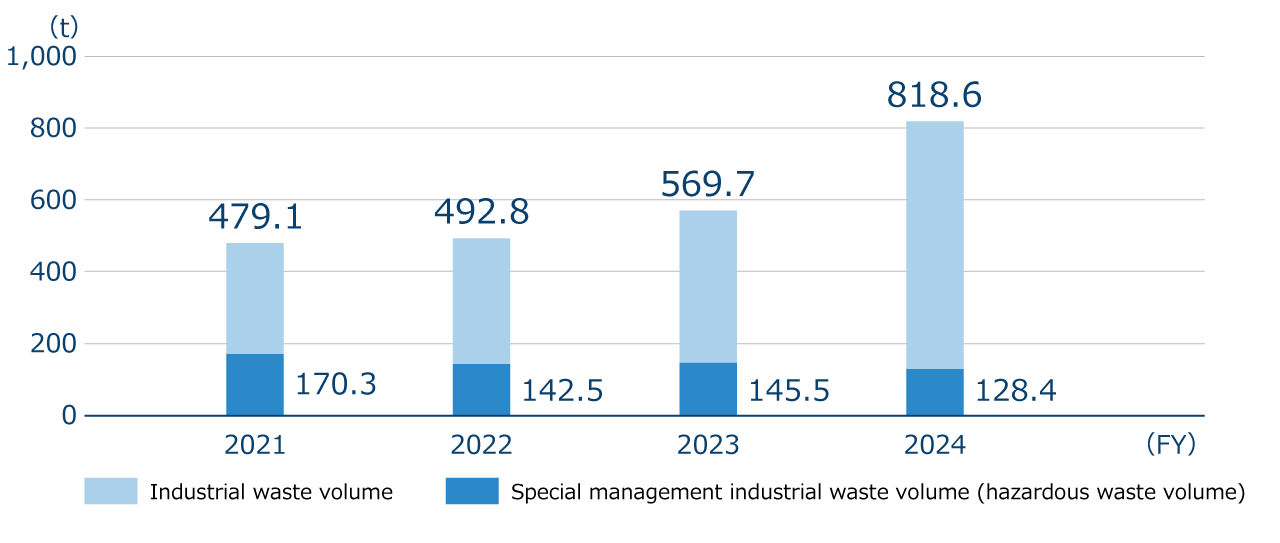
The total volume of our industrial waste in FY2024 was 818.6 tons, a 248.9tons increase compared to the previous fiscal year. This increase was mainly due to (449.4 tons’ worth of) inadequately inactivated experimental wastewater generated at the Tsukuba Research Institute, which was handled as industrial waste after inactivation.
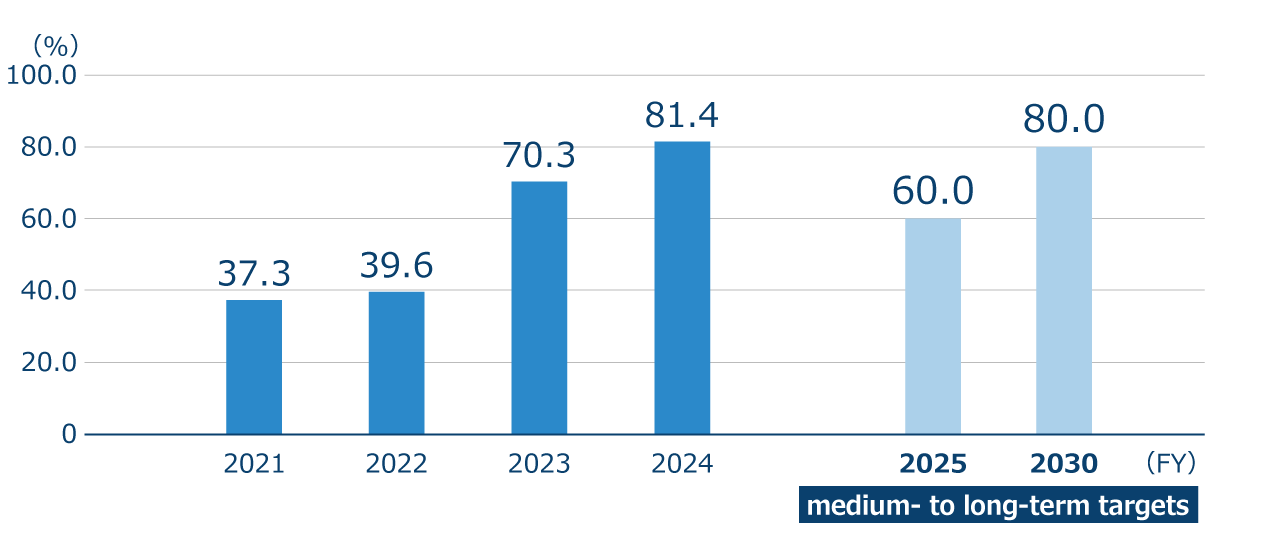
The recycling rate of unnecessary materials (wastes, valuables, free materials, etc.) generated from ONO’s manufacturing plants/research institutes, and logistics centers newly established in FY2022 was 81.4% for FY2024. The main factors behind the results were the continued optimization of industrial waste (including specially controlled industrial waste) treatment contractors and the advancement of other initiatives such as selling experimental equipment that is no longer used with the aim of reuse, as well as promoting the separation of waste. Another main factor behind the results was the fact that experimental wastewater generated at the Tsukuba Research Institute was reused as cooling water for waste incinerators at industrial waste disposal companies after the experimental wastewater had been inactivated. Our calculation method for recycling is based on the Federation of Pharmaceutical Manufacturers’ Associations of Japan’s ideas on recycling, which does not include thermal recycling.
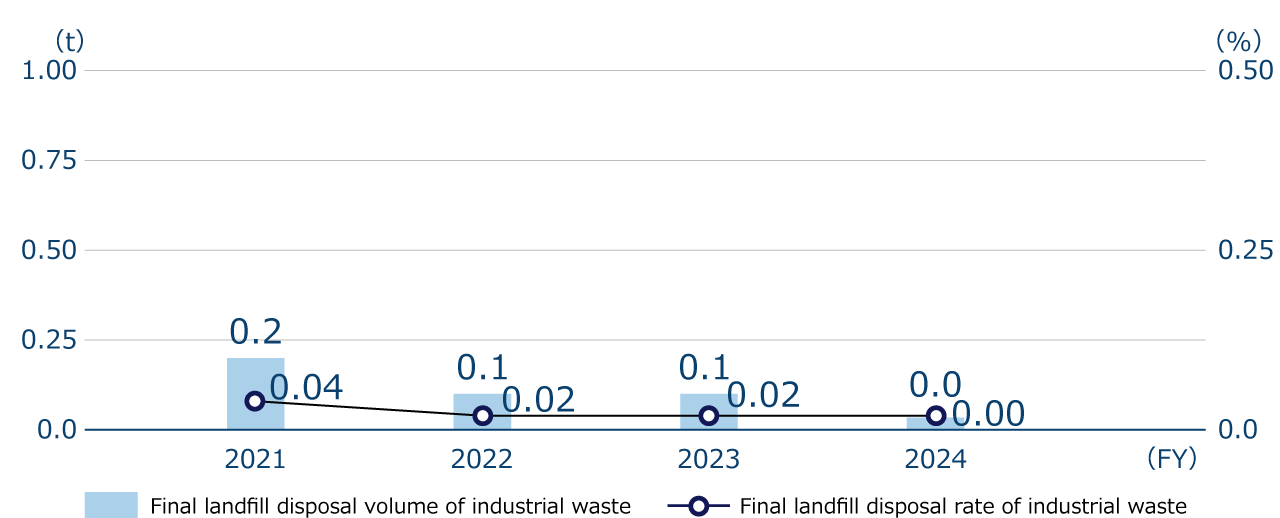
The final landfill rate of our industrial waste in FY2024 was 0.00%. We defined “Reduce the final landfill rate (Final landfill volume/industrial waste volume×100) to no more than 1.0%” as zero emissions. We continued to achieve zero emissions in FY2024 as well by recycling rather than landfilling industrial waste that was emitted in association with business activities.
As for progress management, we clarify issues and initiatives against targets for each site, and conduct self-inspections for environmental data management.
Industrial waste volume and Special management industrial waste volume (hazardous waste volume)
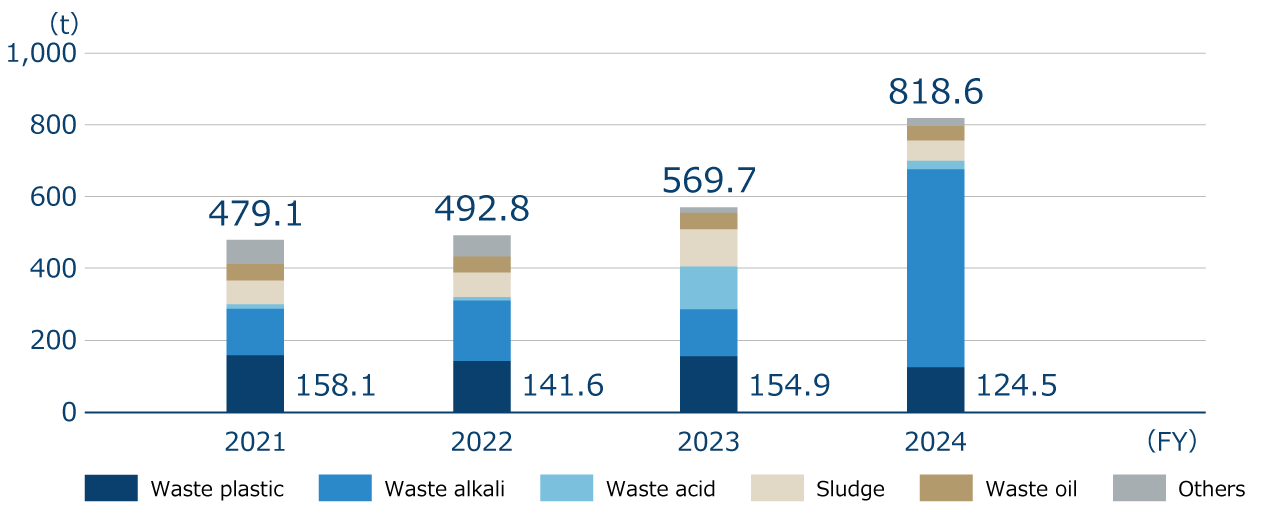
Industrial waste volume (by item)
Final landfill disposal volume and Final landfill disposal rate of industrial waste
Recycling rate
We have organized a nature positive sub-committee (formerly the resource recycling sub-committee) consisting of waste management operators under the Environment Management Committee, and have been promoting activities by setting the following basic policies: "promotion of the 4Rs (Refuse, Reduce, Reuse, and Recycle)" and "selection of materials with a reduced environmental impact.” We have worked to reduce waste generation across the company by reducing paper documents through digitization as well as engaging in the investigation and analysis of processes that generate waste and we are considering and evaluating the introduction of equipment to reduce the volume of waste to reduce emissions. Furthermore, we are promoting resource recycling activities, such as reuse and recycling, etc., and switching materials to materials with low environmental burden.
| Refuse |
|
|---|---|
| Reduce |
|
| Reuse |
|
| Recycle |
|
| Other |
|
In FY2023, the Fujiyama Plant began recycling PTP packaging waste produced during the pharmaceutical packaging process. PTP sheets, which are widely used as a packaging material for pharmaceuticals, are formed through the thermocompression bonding of plastic material and aluminum foil, making them difficult to separate and sort. Unnecessary waste materials could conventionally only be disposed of via incineration, which is why our recycling rate until now has only been at around 10%.
We are now able to recycle 100% as raw material to be used for new products, a feat that we were able to achieve thanks to a peeling machine which separates plastics and aluminum. This also eliminates the need to incinerate packaging waste, thereby reducing our company’s CO2 emissions.

At the Minase Research Institute, kitchen waste and leftover food from cafeterias were incinerated and cinders were disposed of in landfills. However, by taking advantage of special provisions regarding the Waste Disposal Act, etc., in Japan’s Food Recycling Law, the institute has switched to animal feed, which in turn has made it possible to recycle general business waste that is difficult to recycle. In addition, since the number of employees coming to the office to work fluctuates on a daily basis due to the adoption of various work styles such as teleworking, we are striving to reduce food loss by sharing with our cafeteria vendors how many employees are actually coming to the office to work on a daily basis so that appropriate amounts of food can be prepared.

OurNature Positive Subcommittee holds monthly meetings to discuss measures for promoting the 4Rs and proper disposal, to consider initiatives for implementation, and to verify effects, among other matters. In addition, we manage the amount of waste generated at each target site in order to classify waste and identify opportunities for reducing amounts generated, which is shared and promoted by the Nature Positive Subcommittee. In promoting the appropriate disposal of waste, we have determined to give priority to contractors that are certified as excellent companies. On-site observation of intermediate treatment contractors is conducted every year, and we confirm that the appropriate disposal of waste is implemented. The final landfill sites are checked every five years. We continuously implement thorough and appropriate disposal of waste.
We are also working on computer simulation technology in the field of formulation development. This step reduces the number of experiments held and reduces how many raw materials are used (waste).
In addition, we are also working to shift the wet granulation phase of the production process for a portion of our products from a batch production system to a continuous manufacturing system. Doing so will yield various advantages, such as allowing us to respond flexibly to changes in demand while also saving space by making manufacturing equipment more compact. This shift is expected to help reduce the volume of raw materials needed during pharmaceutical development. We estimate that this can allow us to slim down the weight of raw materials needed during pharmaceutical development by approximately 13% when it comes to products under development. By expanding the scope of continuous manufacturing applications in the future, we aim to not only save more energy but also further reduce the volume of raw materials used in our operations.
We strive to extend the validity period of our products by obtaining long-term quality assessment data for each product. Extending the shelf life of products will result in reducing the risk of product disposal due to expiration.
When it comes to product packaging, based on the results of a questionnaire survey at medical institutions on product packaging, we are engaging in activities from the two aspects of ease of use and environmental friendliness. In terms of the environment, we are working to promote the reduction of its environmental impact by changing packaging materials and forms to help save resources and selecting eco-friendly materials. Upon disposal, we have also switched to material labels and packaging forms that encourage recycling.
Furthermore, in response to the results of the questionnaire survey, we changed the method of binding blister package sheets for new products from bag-type (transparent pillow packaging) to band-type. We also changed the tray (partition) of the injection packaging box from plastic to paper materials, which not only reduced the volume of plastic used, but also reduced the capacity (48% per box). With regard to paper consumption, the digitalization of attachments, an initiative which began since August 1, 2021, has reduced paper consumption at our company by approximately 44 tons per year (estimated based on the volume of manufacturing and sales items shipped from our factories in FY2022).
In addition, we have changed paper-based materials for individual packaging boxes to FSC®-certified paper, and switched the inks we use to vegetable oil inks. We also verify the quality of primary packaging that comes into direct contact with pharmaceuticals to further promote the selection of materials that reduce our environmental impact.
| Major initiatives | Progress |
|---|---|
| Changing packaging materials from plastics to paper-based materials | Changing packaging materials for parts of products. Started distribution of the products in FY2020. |
| Reconsideration and changing the method of binding Blister package sheets(Adopting the band-type) | A total of 8 products as of the end of March2025 |
| Switching individual packaging box materials to FSC®-certified paper.* | A total of 38 products as of the end of March2025 |
| Selecting vegetable oil inks. | A total of 14 products as of the end of March2025 |




In Japan, there is an enforced law called the Containers and Packaging Recycling Law, which covers the recycling volume of containers and packaging waste for products sold by sellers. This is to promote the recycling of containers and packaging waste, and based on this law, some of the containers and packaging materials for the products we sell are recycled.
| Container and packaging usage | Obligatory recycling volume | |
|---|---|---|
| Plastic | 207.6 | 60.0 |
| Paper | 163.3 | 0.8 |
| Glass (colorless) | 0.0 | 0.0 |
| Glass (brown) | 0.1 | 0.0 |
| Commissioning fee paid for recycling:3,879 thousand yen | ||
Since FY2024, we have eliminated the use of paper towels in restrooms at major sites where hand dryers are installed. This was done in an aim to promote environmental and forest conservation efforts and to raise employee awareness regarding environmental conservation. We are also working to reduce the use of paper towels at major sites where hand dryers have not been installed, and are gradually installing hand dryers at such sites.
We have introduced paper-based files since January 2020. By switching some plastic files to paper files, we are able to reduce the volume of plastic used.
For photocopies, we perform print management, and a cloud storage system "BOX", which was introduced globally in October 2017, promoted paperless storage and reduced the volume of work required to store and share files. As for purchasing, we have indicated in an easy-to-understand manner whether the products listed in the purchasing system are in compliance with the “Act on Promotion of Procurement of Eco-Friendly Goods and Services by the State and Other Entities” and promoted awareness within us so that each employee has environmental awareness.